Save the date – Clinical Research Governance (CRGo) World Conference & International Clinical Trial Center Network (ICN) Symposium 2023
November 29, 2023 (Wednesday) | 09:30 – 17:00 (GMT+8) | Hong Kong Hybrid Mode |…
Joint Publication
Toward a global harmonization of service infrastructure in academic clinical trial units: an international survey…
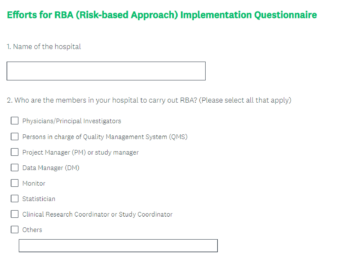
Kyoto University Hospital iACT – Invitation to Participate in RBA Survey
Kyoto University Hospital iACT would like to invite you to participate in this survey, the…
6th International Clinical Trial Center Network (ICN) Steering Board Meeting and Annual General Meeting 2020
HKU-CTC successfully hosted the ICN Steering Board Meeting and Annual General Meeting on 24-25 November,…
ICN New Chair and Vice-chair Elected
We are pleased to announce our new ICN Chair, Henry Yau, and Vice-Chairperson, Christiane Blankenstein….
ICN-ZKS Symposium 2019: Clinical Trials in the Global and Digital World
ZKS Freiburg and ICN annual symposium took place on 27. September 2019 in Freiburg, Germany….
ICN New Chair and Vice-chair Elected
We are pleased to announce the appointment of the new ICN Chairperson, Thomas Hiemstra, and…
Kyoto Research Seeds
The Kyoto University Graduate School of Medicine and Kyoto University Hospital started some years ago…
ICN New Service for Investigators: Consulting for PI
ICN has created a joint service called “Consulting for Principal Investigators”. Participating ICN members offer…
Save the date – Clinical Research Governance (CRGo) World Conference & International Clinical Trial Center Network (ICN) Symposium 2023
November 29, 2023 (Wednesday) | 09:30 – 17:00 (GMT+8) | Hong Kong Hybrid Mode |…
Joint Publication
Toward a global harmonization of service infrastructure in academic clinical trial units: an international survey…
Kyoto University Hospital iACT – Invitation to Participate in RBA Survey
Kyoto University Hospital iACT would like to invite you to participate in this survey, the…
6th International Clinical Trial Center Network (ICN) Steering Board Meeting and Annual General Meeting 2020
HKU-CTC successfully hosted the ICN Steering Board Meeting and Annual General Meeting on 24-25 November,…
ICN New Chair and Vice-chair Elected
We are pleased to announce our new ICN Chair, Henry Yau, and Vice-Chairperson, Christiane Blankenstein….
ICN-ZKS Symposium 2019: Clinical Trials in the Global and Digital World
ZKS Freiburg and ICN annual symposium took place on 27. September 2019 in Freiburg, Germany….
ICN New Chair and Vice-chair Elected
We are pleased to announce the appointment of the new ICN Chairperson, Thomas Hiemstra, and…
Kyoto Research Seeds
The Kyoto University Graduate School of Medicine and Kyoto University Hospital started some years ago…
ICN New Service for Investigators: Consulting for PI
ICN has created a joint service called “Consulting for Principal Investigators”. Participating ICN members offer…